We heard welcome news from NICE recently that a new drug, which effectively treats more genotypes of hepatitis C, has been recommended for use in the NHS.
NICE concluded that the once-daily tablet sofosbuvir-velpatasvir was both clinically and cost-effective, and should be routinely available on the NHS; these preliminary recommendations are now out for public consultation.
So where do we stand on the road to elimination?
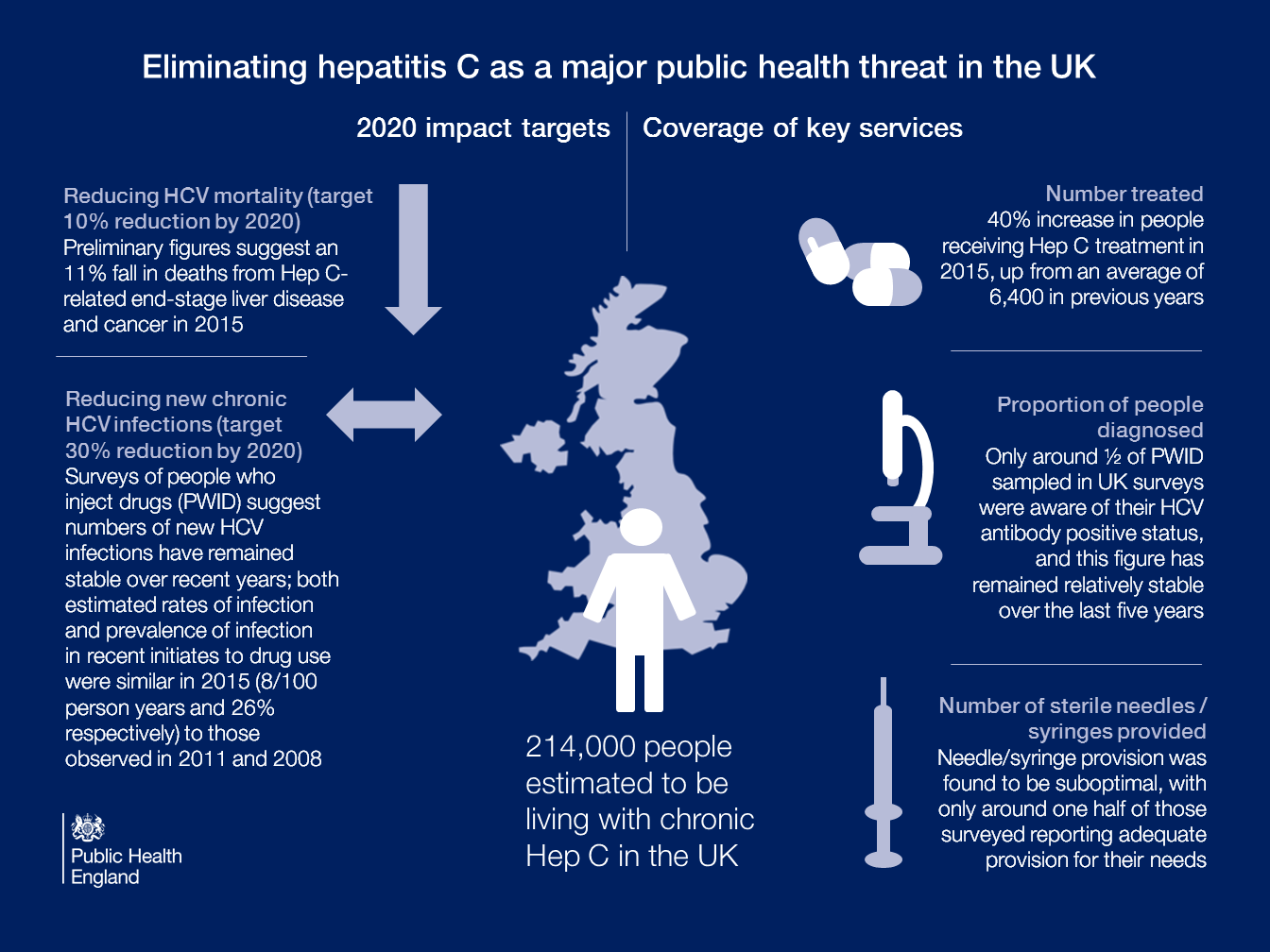
Early indications suggest that improved access to treatment may already be having an impact with preliminary UK figures suggesting a 11% fall in deaths from hepatitis C related end-stage liver disease and cancer in 2015.
This is great news as it is an early indication that, for the first time in over a decade, we may have started to reverse the trend of year on year increases in the number of deaths from severe hepatitis C-related liver disease.
As with other recently approved direct acting antiviral drugs for hepatitis C, the draft guidance recommends that decisions to treat patients with sofosbuvir-velpatasvir are made by multidisciplinary teams in the operational delivery networks put in place by NHS England, to help prioritise treatment for people with the highest unmet clinical need.
But how do we define ‘highest unmet clinical need’? To date, this has quite reasonably led to a focus on prioritising treatment in those with severe disease.
However, we know that this approach comes at a cost, because strategies that focus on treating those with advanced disease have minimal impact on preventing new cases of hepatitis C infection; simply because most new infections in England originate from injecting drug use and this group tend to be in the earlier stages of infection.
So if we are serious about eliminating hepatitis C as a major public health threat, we need to develop strategies that not only save lives by treating those with advanced disease, but also improve and develop our interventions to prevent both new and re-infection.
Modelling studies suggest that treatment of those who are actively injecting, when coupled with other prevention activity, like opioid substitution treatment and needle and syringe programmes, has the potential to reduce the number of new and prevalent infections within this key risk group.
However, unlike recent national data which provides some evidence that treatment may be starting to have an impact on mortality, the data does not show any signs that current prevention strategies are leading to a fall in numbers of new hepatitis C infections, but rather that levels have remained relatively stable.
With treatment strategies initially focused on more advanced disease and national surveys suggesting that only around half of people who inject drugs report adequate needle/syringe provision for their needs, more must be done if we are to reach the WHO Global Health Sector Strategy target to reduce new cases of chronic hepatitis C by 30% by 2020.
Further partnership work is required to extend and develop current treatment and prevention strategies so we can start to drive down the number of new and re-infections.
National data certainly suggest that people who inject drugs are a group with high levels of unmet clinical need.